The Preventer Inhaler

- Floair is a preventer inhaler containing Fluticasone propionate.
- It is available in three strengths 50 mcg, 125 mcg, 250 mcg. Each inhaler has 120 doses.
Floair inhaler reduces swelling and irritation in the air passages in the lungs and helps prevent asthma attacks. Discuss your asthma symptoms with your doctor or pharmacist and ask whether Floair Inhaler is the appropriate choice for you.
To download the Floair Product Brochure and to find out how to use and clean your inhaler, click here.
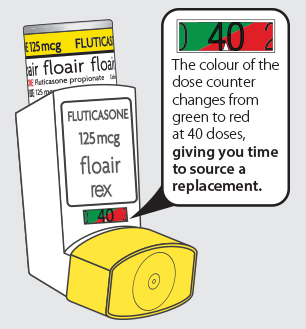
FLOAIR INHALER
HAS A DOSE COUNTER
Floair Inhaler has a dose counter so you don’t need to worry about running out of medication when you need it the most.
Colour of dose counter changes from green to red at 40 doses, giving you time to source a replacement. Monitor your children’s inhalers by checking counter on school days and know you are covered if going away on holiday.
FREQUENTLY ASKED QUESTIONS ABOUT FLOAIR INHALER

Floair Inhaler is a “preventer” inhaler that reduces swelling and irritation in the air passages in your lungs. If you have asthma, taking Floair Inhaler each morning and evening will help prevent asthma attacks. You should always carry your “reliever” inhaler (e.g. Salbutamol) with you.
In the event of an asthma attack, use your Salair Inhaler or your blue “reliever” inhaler, not your Floair Inhaler.
FLOAIR INHALER IS A PRESCRIPTION MEDICINE FOR ORAL INHALATION ONLY.
Floair Inhaler contains Fluticasone propionate as an active medicine. If your previous inhaler also uses the same active medicine i.e. Fluticasone propionate, then Floair Inhaler can be prescribed to manage the same conditions.
Floair Inhaler looks different to other preventer inhalers that you have used. It has a yellow cap and a white body, with a built-in dose counter. The canister with the medicine in it has a yellow and white label.
The first fully funded Fluticasone pMDI with a built-in dose counter in New Zealand.
Floair Inhaler has a built-in dose counter to see how many actuations are left in the inhaler.
After the inhaler is primed for the first time, the dose counter should read 120. This means that there are 120 doses of the medicine left in the inhaler. Each time the inhaler is used, the dose counter will count down by one number.
When there are 40 doses of medicine remaining in the Floair Inhaler, the colour of the dose counter will change from green to red. This gives you time to go to your doctor or pharmacist to pick up another inhaler. You don’t need to worry about running out of medication when you need it the most.
The dose counter will stop counting when it reaches 0. This means there is no medication left in the inhaler and it should be discarded.

If you have previously used another inhaler, you might notice that the spray from Floair Inhaler tastes or feels different in your mouth, or that the inhaler sounds different when sprayed. This does not affect the way your inhaler works.
Discuss your asthma symptoms with your doctor or pharmacist and ask whether Floair Inhaler is the appropriate choice for you.
The Floair Inhaler plastic actuator should be cleaned at least once a week to ensure that it functions correctly.
NEVER wash or soak any part of the inhaler in water.
Use the following technique to clean your inhaler:
- Remove the plastic dust cap from the mouthpiece of the inhaler. The metal canister should NOT be removed from the plastic actuator.
- The plastic mouthpiece and the dust cap are to be wiped inside and outside with a clean dry cloth.
- Replace the plastic dust cap on to the mouthpiece of the inhaler.

Floair inhalers are not funded.
Floair Inhaler with Dose Counter, PRESCRIPTION MEDICINE. Floair Inhaler comes in three different strengths. Floair Inhaler contains either 50 mcg, 125 mcg or 250 mcg of Fluticasone propionate per actuation. Your Floair Inhaler contains 120 actuations. Floair Inhaler is a “preventer” inhaler that reduces irritation and swelling in the air passages in your lungs. The corticosteroid in your Floair Inhaler is not the same anabolic steroids that have been known to have been exploited by sports people. The anti-inflammatory effect in your medication is due to the corticosteroids. You should always carry your “reliever” inhaler (e.g. Salbutamol) with you. In the event of an asthma attack, use your Salair Inhaler or your blue “reliever” inhaler, NOT Floair Inhaler.
Floair Inhaler has risks and benefits. Tell your doctor before using Floair Inhaler if you: are allergic to Fluticasone propionate or any of the other ingredients in your inhaler; have had to discontinue Floair Inhaler in the past or other medication for asthma; have an allergy to dyes, preservatives, foods or other medication; are pregnant or intending on becoming pregnant; are breastfeeding; have been taking or are currently taking steroid tablets or have had a steroid injection; have ever had tuberculosis (TB) or another long-term lung infection; have diabetes; have an infection of the lungs or chest; have heart problems such as heart failure, an irregular heart beat; an abnormal bulging of a blood vessel wall (an aneurysm); high blood pressure; an overactive thyroid gland; low blood levels of potassium; poor adrenal gland function; liver problems; are taking ANY other medicines.
If you experience any of the following side effects, stop taking your Floair Inhaler and seek immediate medical attention as you may be allergic to the medicine: itching, rash or reddened skin; swelling of the eye lids, lips, face or throat; low blood pressure; collapse; increase in wheezing and shortness of breath immediately after using your inhaler. If you experience any of the following side effects advise your nurse, pharmacist or doctor but do not discontinue using your Floair Inhaler: headache; dizziness; an increase in the amount of sugar in your blood; thrush or other fungal infections in the mouth and throat; sinusitis; muscle spasms; coughing or shortness of breath; diarrhoea.
All health providers should be informed that you are using your Floair Inhaler due to the steroids in your medication. Always use Floair Inhaler exactly as your doctor or asthma nurse has told you. Floair Inhaler is for oral inhalation only. It is important that even if you feel that your chest condition has improved, that you do not discontinue using your Floair Inhaler unless your doctor instructs you to. If you have any concerns, discuss them with your doctor or asthma nurse. Discuss your asthma or chronic obstructive pulmonary disease (COPD) symptoms with your doctor or pharmacist and ask whether Floair Inhaler is the appropriate choice for you.
Floair Inhalers are not funded. Other fees – standard doctor’s fees and pharmacy charges – also apply.
Distributed by REX Medical Ltd, Auckland, New Zealand.




